In 1897 he showed that cathode rays (radiation emitted when. Thomson called these particles as negatrons. Thomson managed to estimate its magnitude by performing experiments with charged particles in gases. Experimental results showed that more than 97 wt of lithium in the cathode materials could be selectively recovered with a purity of 99.9 under optimal conditions (FeCl 3:LiFePO 4 mass ratio of 1.2:1, rotary speed of 600 rpm, and reaction time of 30 min. Which clearly indicate that the cathode ray consists of a negatively charged particle. In the mechanochemical process, only ferric chloride was used as a grinding agent. Explain the debate that took place between William Crooke and Heinrich Hertz. The path is deflected by a magnetic field. 
They originate at the cathode and travel to the anode. They move from the negative electrode to the positive electrode. They have energy and momentum, thought to be a property of particles only. Cathode rays also rotate the paddlewheel in their path. It is due to the kinetic energy possessed by the cathode rays. They produce heat energy when they collide with the matter. Importance of Cathode rays in the Study of Atomic Structure: They are used in electron microscopes which are used for a magnifying minute object to the extent that detail of the object can be studied.They are used to find the ratio of charge to mass (e/m) of the electrons.(Cathode Ray Oscillators) which has wide application in electronics. Cathode rays tubes have been developed into C.Shows that cathode rays are made up of negatively charged particles. However, an English physicist J.J Thomson started several different cathode ray experiments in order to find out the ration of the charge to the mass of the. Magnetic field is applied to the path of cathode rays, they are deflected. If discharge tube is highly evacuated so that the pressure in the tube is of the order of 0.01 mm of mercury, certain rays are emitted by the cathode in the form of bluish streamers. Separation of Li and Co from LiCoO2 Cathode Material Through Aluminothermic Reduction Using Different Aluminum Sources: Chemical Grade, Swarf, and Dross February 2023 DOI: 10. This study of the discharge can be done at different pressures. 
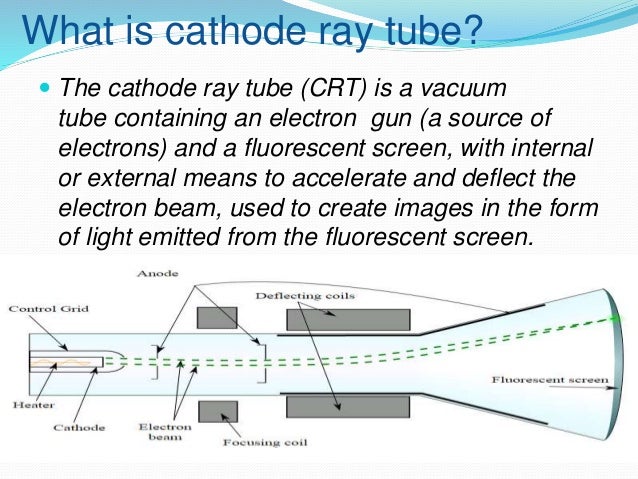
The pressure can be read from the pressure gauge and can be made constant. The tube is filled with air or any other gas in which the electric discharge at different low pressures is to be studied.Īs the vacuum pump is operated the pressure of the gas in the discharge tube is gradually reduced and the discharge goes through different stages. summarizing the conclusions from the gold foil experiment as a theory. On a soda glass screen put in the tube, they produce a greenish-yellow glow. Cathode ray tubes consist of an evacuated glass with two metal electrodes and rarefied gas within the. They were previously called gas discharge tubes or Crookes tubes (named after William Crookes whose experiments showed t he earliest direct signs of electrons and their charge). Phosphorescent materials are the only way to see these rays. Nowadays, we use the name cathode ray tubes. Cathode rays originate at the cathode and move towards the anode. Experimental results showed that more than 97 wt of lithium in the cathode materials could be selectively recovered with a purity of 99.9 under optimal conditions (FeCl 3 :LiFePO 4 mass ratio of 1. voltage of 10,000 to 15,000 volts can be applied between the electrodes. cathode ray particles can move at very fast speeds. The observations of the cathode ray experiment are as follows- 1. Electrodes are disc-shaped made up of aluminium are sealed in the tube at two ends. Discovery of the Electron: Cathode Ray Tube Experiment Tyler DeWitt 1. The tube is closed at both ends and has a side tube connected to a vacuum pump and a pressure gauge. The discharge tube consists of a thick-walled glass tube about 40 to 50 cm in length and about 3 to 4 cm in diameter. Their properties are independent from the electrodes and gas present in the cathode ray tube.The Discharge tube is a glass tube used to study the flow of electricity through gases at low pressures.They travel from the cathode to the anode.Charge/mass ratio of the rays is constant.Cathode ray tubes (CRTs) use a focused beam of electrons deflected by electric or magnetic fields to create the image in a classic television set. He showed the rays were composed of a previously unknown negatively charged particle, which was later called the electron. Įlectrons were discovered as the constituents of cathode rays in 1897 by British physicist J. They were first observed in 1869 by German physicist Johann Hittorf, and were named in 1876 by Eugen Goldstein Kathodenstrahlen, or cathode rays. A cathode ray tube aims the ray at a point on its screen. It is called a "cathode ray" because the electrons are being emitted from the negative charged element in the vacuum tube called the cathode. A cathode ray is a stream of electrons that are seen in vacuum tubes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
